Past Projects
Aryl-ethynyl Sensors
Many anions are problematic environmental contaminants and are vital to many processes in nature, with anion binding proteins and transport channels implicated in the mechanisms of many disease pathways. This avenue of research is a productive collaboration between the Haley lab and that of UO colleague Darren Johnson to target new organic receptors that selectively bind and sense anions. The project is a union of the synthetic expertise of the Haley group toward the assembly of relatively rigid and inherently fluorescent molecules based on arylethynyl scaffolding with the extensive knowledge of the Johnson lab to design and exhaustively analyze complex supramolecular systems.


The modularity of our design strategy allows for exploration of a variety of recognition motifs for anions, including electrostatic attractions, hydrogen bond interactions and attractions with electron-deficient arenes (anion-π, CH•••X– hydrogen bonds and weak-sigma complexes). This flexibility affords the possibility of selectively binding anions that are challenging to target with traditional approaches. Core and linker substitution provides another approach to tuning the selectivity of the receptors by changing the shape and size of the binding pocket, in addition to modification of the fluorescent behavior through adjustment of the electronics of the conjugated core fluorophore. The functionality of the receptors can also be adjusted to provide water-solubility to the molecules or even cell membrane permeability. These sensors will have long-term applications in sensing, imaging and/or remediating anions, which will impact public health in both discovering and removing environmental contaminants and imaging the role anions play in biological processes.
Relevant Publications
(1) Fargher, H. A.; Delmau, L. H.; Bryantsev, V. S.; Haley, M. M.; Johnson, D. W.; Moyer, B. A. Chem. Sci. 2024, 15, 5311-5318.
(2) Fargher, H. A.; Sherbow, T. J.; Haley, M. M.; Johnson, D. W.; Pluth, M. D. Chem. Soc. Rev. 2022, 51, 1451-1469.
(3) Fargher, H. A.; Nickels, R. A.; de Faria, T. P.; Haley, M. M.; Pluth, M. D.; Johnson, D. W. RSC Adv. 2021, 11, 26581-26585.
(4) Sherbow, T. J.; Fargher, H. A.; Haley, M. M.; Johnson, D. W. J. Org. Chem. 2020, 85 12367-12373.
(5) Fargher, H. A.; Lau, N.; Richardson, H. C.; Cheong, P. H.-Y.; Haley, M. M.; Pluth, M. D.; Johnson, D. W. J. Am. Chem. Soc. 2020, 142, 8243-8251.
(6) Eytel, L. M.; Fargher, H. A.; Haley, M. M.; Johnson, D. W. Chem. Commun. 2020, 55, 5195-5206.
(7) Lohrman, J. A.; Deng, C.-L.; Shear, T. A.; Zakharov, L. N.; Haley, M. M.; Johnson, D. W. Chem. Commun. 2019, 55, 1919-1922.
(8) Fargher, H. A.; Lau, N.; Zakharov, L. N.; Haley, M. M.; Johnson, D. W.; Pluth, M. D. Chem. Sci. 2019, 10, 67-72.
(9) Eytel, L. M.; Brueckner, A. C.; Lohrman, J. A.; Haley, M. M.; Cheong, P. H.-Y.; Johnson, D. W. Chem. Commun. 2018, 54, 13208-13211.
(10) Tresca, B. W.; Brueckner, A. C.; Haley, M. M.; Cheong, P. H.-Y.; Johnson, D. W. J. Am. Chem. Soc. 2017, 139, 3962-3965.
(11) Eytel, L. M.; Gilbert, A. K.; Gorner, P.; Zakharov, L. N.; Johnson, D. W.; Haley, M. M. Chem. Eur. J. 2017, 23, 4051-4054.
(12) Hartle, M. D.; Hansen, R. J.; Tresca, B. W.; Prakel, S. S.; Zakharov, L. N.; Haley, M. M.; Pluth, M. D.; Johnson, D. W. Angew, Chem. Int. Ed. 2016, 55, 11480-11484.
(13) Vonnegut, C. L.; Shonkwiler, A. M.; Zakharov, L. N.; Haley, M. M.; Johnson, D. W. Chem. Commun. 2016, 52, 9506-9509.
(14) Tresca, B. W.; Berryman, O. B.; Zakharov, L. N.; Johnson, D. W.; Haley, M. M. Supramol. Chem. 2016, 28, 37-44.
(15) Tresca, B. W.; Hansen, R. J.; Chau, C. V.; Hay, B. P.; Zakharov, L. N.; Haley, M. M.; Johnson, D. W. J. Am. Chem. Soc. 2015, 137, 14959-14967.
(16) Vonnegut, C. L.; Shonkwiler, A. M.; Khalifa, M. K.; Zakharov, L. N.; Johnson, D. W.; Haley, M. M. Angew. Chem. Int. Ed. 2015, 54, 13318-13322.
(17) Watt, M. M.; Engle, J. M.; Fairley, K. C.; Robitshek, T. E.; Haley, M. M.; Johnson, D. W. Org. Biomol. Chem. 2015, 13, 4266-4270.
(18) Berryman, O. B.; Johnson, C. A.; Vonnegut, C. L.; Fajardo, K. A.; Zakharov, L. N.; Johnson, D. W.; Haley, M. M. Cryst. Growth Des. 2015, 15, 1502-1511.
(19) Vonnegut, C. L.; Tresca, B. W.; Johnson, D. W.; Haley, M. M. Chem. Asian J. 2015, 10, 522-535.
(20) Gavette, J. V.; Evoniuk, C. J.; Zakharov, L. N.; Carnes, M. E.; Haley, M. M.; Johnson, D. W. Chem. Sci. 2014, 5, 2899–2905.
(21) Gavette, J. V.; Klug, C. M.; Zakharov, L. N.; Shores, M. P.; Haley, M. M.; Johnson, D. W. Chem. Commun. 2014, 50, 7173–7175.
(22) Engle, J.M.; Singh, P.S.; Vonnegut, C.L.; Zakharov, L.N.; Johnson, D.W.; Haley, M.M. CrystEngComm 2014, 16, 3703-3706.
(23) Watt, M. M.; Zakharov, L. N.; Haley, M. M.; Johnson, D. W. Angew. Chem. Int. Ed. 2013, 52, 10275–10280.
(24) Gavette, J. V.; Mills, N. S.; Zakharov, L. N.; Johnson, C. A.; Johnson, D. W.; Haley, M. M. Angew. Chem. Int. Ed 2013, 52, 10460–10464.
(25) Tresca, B. W.; Zakharov, L. N.; Carroll, C. N.; Johnson, D. W.; Haley, M. M. Chem. Commun. 2013, 49, 7240-7242.
(26) Engle, J. M.; Carroll, C. N.; Johnson, D. W.; Haley, M. M. Chem. Sci. 2012, 3, 1105-1110.
(27) Engle, J. M.; Lakshminarayanan, P. S.; Carroll, C. N.; Zakharov, L. N.; Haley, M. M.; Johnson, D. W. Cryst. Growth Des. 2011, 11, 5144-5152.
(28) Gavette, J. V.; Lara, J.; Berryman, O. B.; Zakharov, L. N.; Haley, M. M.; Johnson, D. W. Chem. Commun. 2011, 47, 7653-7655.
(29) Carroll, C. N.; Coombs, B. A.; McClintock, S. P.; Johnson II, C. A.; Berryman, O. B.; Johnson, D. W.; Haley, M. M. Chem. Commun. 2011, 47, 5539-5541.
(30) Carroll, C. N.; Naleway, J. J.; Haley, M. M.; Johnson, D. W. Chem. Soc. Rev. 2010, 39, 3875–3888.
(31) Johnson, C. A.; Berryman, O. B.; Sather, A. C.; Zakharov, L. N.; Haley, M. M.; Johnson , D. W. Cryst. Growth Des., 2009, 9, 4247–4249.
(32) Carroll, C. N.; Berryman, O. B.; Johnson, C. A.; Zakharov, L. N.; Haley, M. M.; Johnson, D. W. Chem. Commun. 2009, 18, 2520–2522.
(33) Berryman, O. B.; Johnson, C. A.; Zakharov, L. N.; Haley, M. M.; Johnson D. W. Angew. Chem. Int. Ed. 2008, 47, 117-120.
Aryl-ethynyl Frameworks
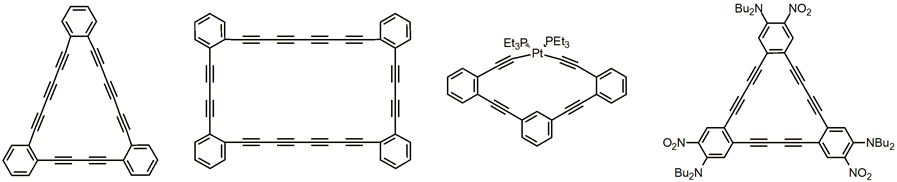
Carbon-rich materials are of extreme interest to researchers in many fields, and have become the subject of an increasing number of experimental and theoretical studies due to the isolation and characterization of the fullerenes (C60, C70, etc.) and now graphene in macroscopic quantities. The major focus of our research into these materials was on two overlapping subsets: (a) carbon networks and models, and (b) molecules with a high C:H ratio. Both studies are based on a class of molecules known as dehydrobenzoannulenes (DBAs).
Calculations predict stable, low energy phases of carbon consisting of stacked, planar carbon layers occupied by sp and sp2 states. The properties of these novel carbon networks are of great relevance in the search for organic conductors, electrochromic display materials, liquid crystals, synthetic ferromagnets, and non-linear optical substances. We have prepared a variety of DBA model compounds for each network in order to compare the chemical and physical properties of the models with those we observe for the corresponding polymeric materials, thus allowing us to probe the monomer/polymer interface.
New synthetic methods developed during the network studies have allowed us to assemble a diverse array of DBA topologies, with nearly 100 macrocycles completed. The structures illustrated below represent some of these molecules, including the first examples of DBAs containing more than two consecutive acetylenic units per side. We also prepared structures incorporating transition metal fragments. Most importantly, the stepwise assembly process used to synthesize our macrocycles allows us to tailor the substituent placement on the aromatic rings, creating for the first time derivatized donor-acceptor structures for nonlinear optical applications.
Relevant Publications
(1) Sandoval, J. S.; Haley, M. M.; Goodson III, T. J. Phys. Chem. A 2025, 129, 1240-1251.
(1) Nojo, W.; Reingold, I. D.; Bard, J. P.; Chase, D. T.; Deng, C.-L.; Haley, M. M. ChemPlusChem 2019, 84, 1391-1395.
(2) Takeda, T.; Haley, M. M. Can. J. Chem., 2017, 138, 16827-16838.
(3) O’Connor, M. J.; Yelle, R. B.; Linz, T. M.; Haley, M. M. Comptes Rendus Chimie 2009, 12, 385–394.
(4) Spitler, E. L.; Haley, M. M. Tetrahedron 2008, 64, 11469–11474.
(5) O’Connor, M. J.; Haley, M. M. Org. Lett. 2008, 10, 3973–3976.
(6) O’Connor, M. J.; Yelle, R. B.; Zakharov, L. N.; Haley, M. M. J. Org. Chem. 2008, 73, 4424–4432.
(7) Samori, S.; Tojo, S.; Fujitsuka, M.; Spitler, E. L.; Haley, M. M.; Majima, T. J. Org. Chem. 2008, 73, 3551–3558.
(8) Spitler, E. L.; Haley, M. M. Org. Biomol. Chem. 2008, 6, 1569–1576.
(9) Spitler, E. L.; Monson, J. M.; Haley, M. M. J. Org. Chem. 2008, 73, 2211–2223.
(10) Tahara, K.; Johnson, C. A.; Fujita, T.; Sonoda, M.; De Schryver, F. C.; De Feyter, S.; Haley, M. M.; Tobe, Y. Langmuir 2007, 23, 10190–10197.
(11) Johnson, C. A.; Lu, Y.; Haley, M. M. Org. Lett. 2007, 9, 3725–3728.
(12) Spitler, E. L.; McClintock, S. P.; Haley, M. M. J. Org. Chem. 2007, 72, 6692–6699.
(13) Samori, S.; Tojo, S.; Fujitsuka, M.; Spitler, E. L.; Haley, M. M.; Majima, T. J. Org. Chem. 2007, 72, 2785–2793.
(14) Spitler, E. L.; Shirtcliff, L. D.; Haley, M. M. J. Org. Chem. 2007, 72, 86–96.
(15) Spitler, E. L.; Johnson, C. A.; Haley, M. M. Chem. Rev. 2006, 106, 5344–5386.
(16) Slepkov, A. D.; Hegmann, F. A.; Tykwinski, R. R.; Kamada, K.; Ohta, K.; Marsden, J. A.; Spitler, E. L.; Miller, J. J.; Haley, M. M. Opt. Lett. 2006, 31, 3315–3317.
(17) Bhaskar, A.; Guda, R.; Haley, M. M.; Goodson J. Am. Chem. Soc. 2006, 128, 13972–13973.
(18) Anand, S.; Varnavski, O.; Marsden, J. A.; Haley, M. M.; Schlegel, H. B.; Goodson, T. J. Phys. Chem. A 2006, 110, 1305–1318.
(19) Hinrichs, H.; Boydston, A. J.; Jones, P. G.; Hess, K.; Herges, R.; Haley, M. M.; Hopf, H. Chem. Eur. J. 2006, 12, 7103–7115.
(20) Marsden, J. A.; Haley, M. M. J. Org. Chem. 2005, 70, 10213–10226.
(21) Hinrichs, H.; Fischer, A. K.; Jones, P. G.; Hopf, H.; Haley, M. M. Org. Lett. 2005, 7, 3793–3795.
(22) Marsden, J. A.; Miller, J. J.; Shirtcliff, L. D.; Haley, M. M. J. Am. Chem. Soc. 2005, 127, 2464–2476.
(23) Johnson, C. A.; Haley, M. M.; Rather, E.; Han, F.; Weakley, T. J. R. Organometallics 2005, 24, 1161–1172.
(24) Marsden, J. A.; O’Connor, M. J.; Haley, M. M. Org. Lett. 2004, 6, 2385–2388.
Coarctate Cyclizations
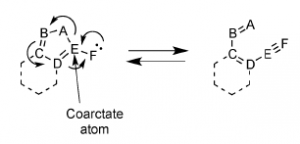
Coarctate reactions can be identified by so-called “coarctations”, an atom or linear sequence of atoms in which two bonds are broken and two bonds are formed in a single step. These coarctations are bound by “terminators”, which consist of an atom with a lone pair, two atoms complementing the electron shift to a three-membered ring, four atoms complementing the shift to a five-membered ring, etc.
In a paper published by Rainer Herges in 1994 
(DOI:10.1021/ci00017a011) 74 conceivable reactions of the latter type were listed. Some of these reactions were known, most were predicted as possible candidates for future discovery. Our isoindazole cyclization turned out to fit reaction #71 in the list; thus, a new coarctate reaction was found, and explored within our lab for several years as a collaboration with The Herges Lab.
Relevant Publications
(1) Young, B. S.; Herges, R.; Haley, M. M. Chem. Commun. 2012, 48, 9441–9455.
(2) Young, B. S.; Marshall, J. L.; MacDonald, E.; Vonnegut, C. L.; Haley, M. M. Chem. Commun. 2012, 48, 5166–5168.
(3) Young, B. S.; Köhler, F.; Herges, R.; Haley, M. M. J. Org. Chem. 2011, 76, 8483–8487.
(4) McClintock, S. P.; Zakharov, L. N.; Herges, R.; Haley, M. M. Chem. Eur. J. 2011, 17, 6798–6806.
(5) McClintock, S. P.; Shirtcliff, L. D.; Herges, R.; Haley, M. M. J. Org. Chem. 2008, 73, 8755–8762.
(6) Shirtcliff, L. D.; Rivers, J.; Haley, M. M. J. Org. Chem. 2006, 71, 6619–6622.
(7) Shirtcliff, L. D.; Weakley, T. J. R.; Haley, M. M.; Köhler, F.; Herges, R. J. Org. Chem. 2004, 69, 6979–6985.
(8) Kimball, D. B.; Weakley, T. J. R.; Herges, R.; Haley, M. M. J. Am. Chem. Soc. 2002, 124, 13463–13473.
(9) Kimball, D. B.; Herges, R.; Haley, M. M. J. Am. Chem. Soc. 2002, 124, 1572–1573.
(10) Kimball, D. B.; Haley, M. M. Angew. Chem. Int. Ed. 2002, 41, 3338–3351.
(11) Kimball, D. B.; Hayes, A. G.; Haley, M. M. Org. Lett. 2000, 2, 3825–3827.
Metallabenzenes
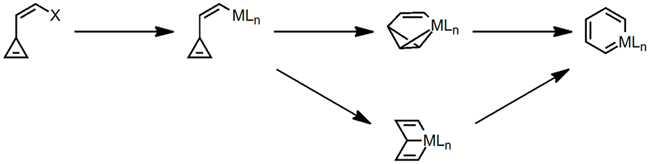
This area of chemistry pushes the frontiers of our understanding of the bonding and reactivity in organometallics as often time systems of this type defy conventional wisdom. The key to our research is the preparation of cyclopropenes containing additional unsaturated moieties and their subsequent reaction with transition-metal reagents to yield novel organometallic complexes.
A metallabenzene is a transition-metal analog of benzene in which one methine (CH) is replaced by an isoelectronic MLn fragment, yet the molecule retains “aromatic” physical and chemical properties. Although two dozen or so such structures were known when we started this project, there was no general route that allowed entry into this class of molecules. Suitably substituted 3-vinylcyclopropenes made a general route accessible due to the inherent reactivity of the strained cyclopropene to undergo ring cleavage or ring expansion. Depending upon the substitution pattern on the cyclopropene and the organometallic reagent used, it became possible to isolate the corresponding metallabenzene valence isomers.
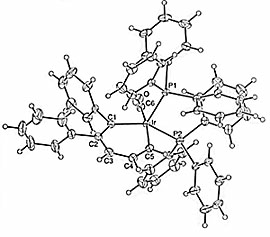
Using Vaska complexes and a 1,2-diphenyl cyclopropene derivative, we have prepared several new “iridabenzenes” as well as the first examples of “iridabenzvalenes” (Figure 1). Like a normal benzene valence isomer, iridabenzvalene cleanly rearranges to an iridabenzene upon heating.
 |
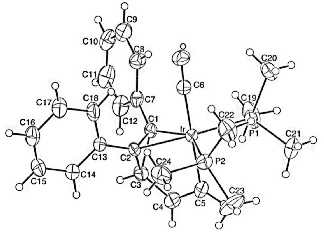 |
Figure 1. Molecular structures of iridabenzene and iridabenzvalene, respectively; ellipsoids drawn at the 30% level.
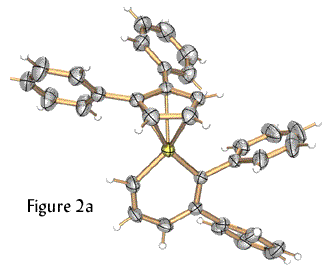
We extended this new methodology for metallabenzenes to utilize other metals. The formation of a platinabenzene and a rhodabenzvalene, which are shown in Figure 2, illustrate the diversity of our route.
 |
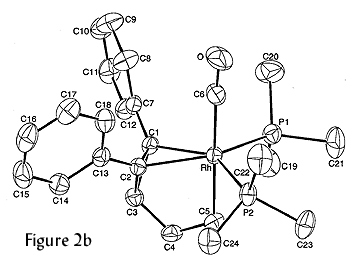 |
Figure 2. Molecular structures of a platinabenzene (left, 2a) and a rhodabenzvalene (right, 2b).
Relevant Publications
(1) Haley, M.M. “Metallabenzenes and Valence Isomers via the Nucleophilic 3-Vinylcyclopropene Route” In Metallabenzenes: An Expert View; Wright, L.J. (Ed.); Wiley: Hoboken, NJ, 2017, pp 109-143.
(2) Jacob, V.; Landorf, C. W.; Zakharov, L. N.; Weakley, T. J. R.; Haley, M. M. Organometallics 2009, 28, 5183–5190.
(3) Wu, H.-P.; Ess, D. H.; Lanza, S.; Weakley, T. J. R.; Houk, K. N.; Baldridge, K. K.; Haley, M. M. Organometallics 2007, 26, 3957–3968.
(4) Johnson II, C. A.; Baker, B. A.; Berryman, O. B.; Zakharov, L. N.; O’Connor, M. J.; Haley, M. M. J. Organomet. Chem. 2006, 691, 413–421.
(5) Landorf, C. W.; Haley, M. M. Ang. Chem. Int. Ed. 2006, 45, 3914–3936.
(6) Wu, H.-P.; Weakley, T. J. R.; Haley, M. M. Chem. Eur. J. 2005, 11, 1191–1200.
Myotonic Dystrophy
In a close collaboration with former UO biochemist Andy Berglund and his lab, we developed therapeutics for Myotonic Dystrophy (DM). DM is the most common form of adult onset muscular dystrophy, affecting 1 in 8000 people. DM is caused by an expansion of three or four nucleotide repeats–CUG expansions in patients with myotonic dystrophy type 1 (DM1), and CCUG expansions in patients with myotonic dystrophy type 2 (DM2). Both DM1 and DM2 patients display many of the same symptoms (myotonia, progressive muscle weakness and wasting, cataracts, cardiac and developmental defects) strongly suggesting that both DM1 and DM2 progress through the same or very similar mechanisms.
In 2007 the Berglund lab discovered that pentamidine (1) will preferentially bind CUG repeats over other nucleotides and help to reverse some of the molecular defects of DM. We prepared a wide variety of pentamidine analogues (2-4) for determining structure-activity relationships to improve efficacy and decrease toxicity versus 1 and thus potentially generate a therapeutic for DM, in addition to providing additional tools to aid in the understanding of the roles of CUG repeats in DM progression.
Relevant Publications
(1) Khalifa, M. K.; Bodner, M. J.; Berglund, J. A.; Haley, M. M. Tetrahedron Lett. 2015, 56, 4109-4111.
(2) Siboni, R. B.; Bodner, M. J.; Khalifa, M. J.; Docter, A. G.; Choi, J. Y.; Nakamuri, M.; Haley, M. M.; Berglund, J. A. J. Med. Chem. 2015, 58, 5770-5780.
(3) Coonrod, L. A.; Nakamori, M.; Wang, W.; Carrell, S.; Hilton, C. L.; Bodner, M. J.; Siboni, R. B.; Docter, A. G.; Haley, M. M.; Thornton, C.; Berglund, J. A. ACS Chem. Biol. 2013, 8, 2528-2537.
(4) Warf, M. B.; Nakamori, M.; Matthys, C. M.; Thornton, C. A.; Berglund, J. A. Proc. Nat. Ac. Sci. 2009, 106, 18551–18556.
Pt-based Coordination Complexes
In collaboration with the DeRose lab
This collaboration successfully combines the synthetic expertise of the Haley lab with the demonstrated Pt-RNA biochemistry knowledge of UO colleague Victoria DeRose’s lab. The goal of this research is to use chemical biology approaches to identify interactions between platinum-based reagents and cellular constituents. We are developing Pt(II) analogues that are synthetically modified for post-treatment “click”-chemistry, and use these compounds for identifying binding partners and cellular localization. Haley serves as the synthetic advisor/expert for the preparation of the organic ligands and of the complex fluorophores that are subsequently bound/”clicked” to Pt. 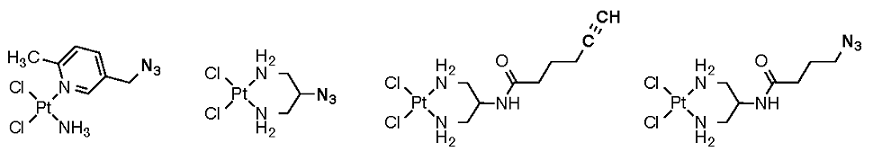
Relevant Publications
1)White, J. D.; Haley, M. M.; DeRose, V. J. Acc. Chem. Res. 2016, 49, 56-66.
DOI: 10.1021/acs.accounts.5b00322
2) Wirth, R.; White, J. D.; Moghaddam, A. D.; Ginzburg, A. L.; Zakharov, L. N.; Haley, M. M.; DeRose, V. J. . J. Am. Chem. Soc. 2015, 137, 15169-15175.
DOI: 10.1021/jacs.5b09108
3) Moghaddam, A. D.; White, J. D.; Cunningham, R. N.; Loes, A. N.; Haley, M. M.; DeRose, V. J. Dalton Trans. 2015, 44, 3536-3539.
DOI: 10.1039/C4DT02649G
4) White, J. D.; Guzman, L. E.; Zakharov, L. N.; Haley, M. M.; DeRose, V. J. Angew. Chem. Int. Ed. 2015, 54, 1032-1035.
DOI: 10.1002/anie.201409853
5) Osborn, M. F.; White, J. D.; Haley, M. M.; DeRose, V. J. ACS Chem. Biol. 2014, 9, 2404-2411.
DOI: 10.1021/cb500395z
6) White, J. D.; Osborn, M. F.; Moghaddam, A. D.; Guzman, L. E.; Haley, M. M.; DeRose, V. J. J. Am. Chem. Soc. 2013, 135, 11680-11683.
DOI: 10.1021/ja402453k